Mixed Lymphocyte Reaction (MLR)
Figure 1: One-Way MLR Mechanism
Schematic illustration of the one-way allogeneic mixed lymphocyte reaction (MLR) experimental workflow. Cord blood (CB) derived dendritic cells (DCs) from Donor 1 were co-cultured with mononuclear cells (MNCs) from Donor 2 and exposed to the test compound. Antigen presentation through the major histocompatibility complex (MHC)–T cell receptor (TCR) interaction resulted in T-cell activation. T-cell activation was assessed by surface expression of early (CD69) and late (CD25) activation markers using flow cytometry, along with functional readouts including T-cell proliferation and cytokine secretion. Pro-inflammatory cytokines, including IFN-γ and TNF-α, were quantified by ELISA.
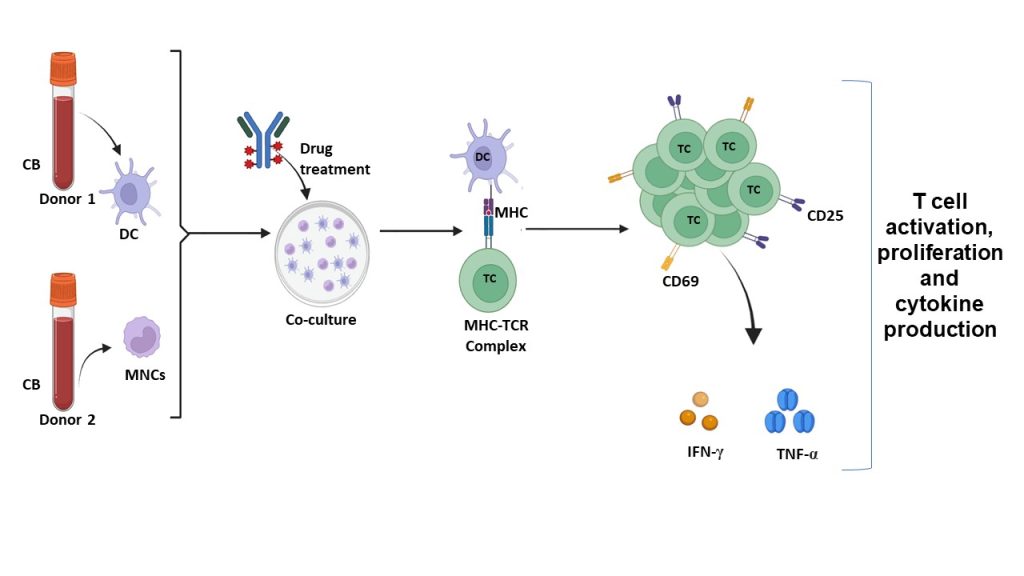
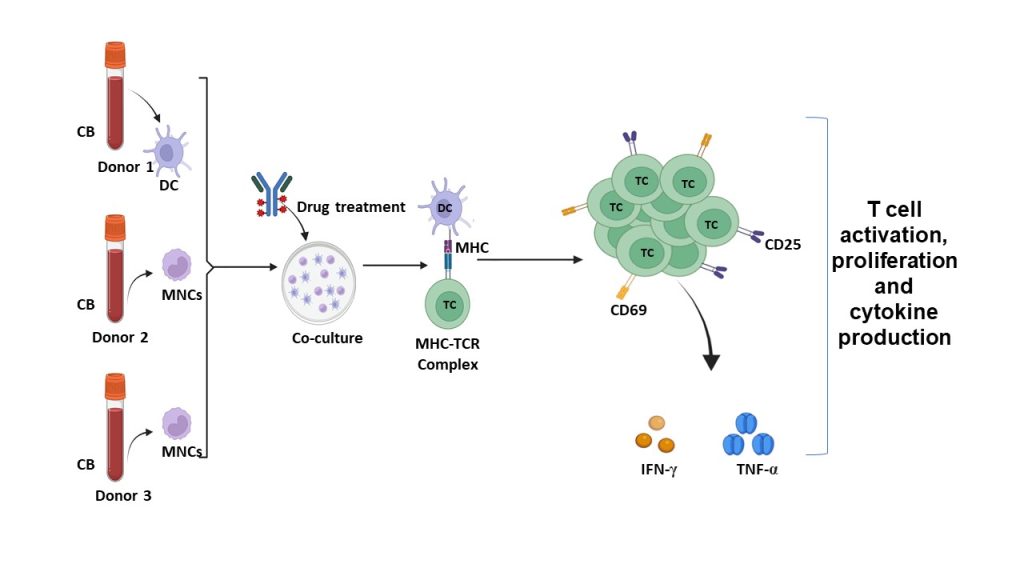
Figure 2: Two-Way MLR Mechanism
Schematic illustration of the one-way allogeneic mixed lymphocyte reaction (MLR) experimental workflow. Cord blood (CB) derived dendritic cells (DCs) from Donor 1 were co-cultured with mononuclear cells (MNCs) from Donor 2 and Donor 3 and exposed to the test compound. Antigen presentation through the major histocompatibility complex (MHC)–T cell receptor (TCR) interaction resulted in T-cell activation. T-cell activation was assessed by surface expression of early (CD69) and late (CD25) activation markers using flow cytometry, along with functional readouts including T-cell proliferation and cytokine secretion. Pro-inflammatory cytokines, including IFN-γ and TNF-α, were quantified by ELISA.
Introducing EFIKC-MLR
EFIKC-MLR harnesses the power of agentic AI, integrated into an in vitro immunological milieu (biological model), to revolutionize the way you compute drug relative potency while predicting cytokine expression—ensuring you can effectively authenticate drug effects.

Key Features
-
Offers customizable, tailored workflows that adapt to your specific study design
-
Real-time analysis of relative potency in minutes owing to the optimisations and the embedded biological model
-
User-friendly, Simple, yet Robust workflow
-
AI-Enabled Analysis
Leverage advanced AI algorithms to deliver accurate and reliable potency assessments
-
Comprehensive Data Insights
Utilize drug-treated human immune microphysiological system data points for enhanced analysis
-
Enterprise Solution leveraging human in vitro microphysiological platform: Seamlessly integrate into your existing workflows for optimal efficiency and scalability
Why Choose EFIKC-MLR?
Streamlined Drug Development
Accelerate your product development timelines with precise data interpretation from the modelled human immunological system.
Enhanced Predictive Capabilities
Gain deeper insights into cytokine profiles, guiding you in decision-making processes.
Regulatory Compliance
Stay ahead of the curve with a solution designed to meet FDA standards.
Why EFIKC-MLR?
-
We are the only group that has the Agentic AI-enabled MLR Bioassay offered as an enterprise solution, leveraging human microphysiologies, to the global industry
-
It is a tried and tested solution, ready to adopt
-
Transform your drug potency testing with EFIKC-MLR and unlock the true potential of your bio-pharmaceutical innovations.

